Welcome To My Website....
A little about MYSELF
#worlddrugtracker #helpingmillions #opensuperstar #amcrasto #newdrugapprovals #90percentparalysis #onwheelchopper #hotpersoninawheelchair
ι мαкє мy σωη яυℓєร, мy σωη вσυη∂αяιєร, мy σωη ℓιмιт
ι мαкє мy σωη яυℓєร, мy σωη вσυη∂αяιєร, мy σωη ℓιмιт

16 JAN 2023
LINK
Dr. Anthony Melvin Crasto, graduated from Mumbai University, Completed his Ph.D from ICT, 1991, Mumbai, India, in the field of Organic Chemistry, Currently he is working with AFRICURE PHARMA, ROW2TECH, NIPER-G, Department of Pharmaceuticals, Ministry of Chemicals & Fertilizers, Govt. of India as ADVISOR from Feb 2023, earlier assignment was Consultant GLENMARK LIFESCIENCES LTD JAN 2022-23, Retired from Glenmark in JAN 2022 [DEC05 JOINED ]as Principal Scientist, in Research centre Process Research at Mahape, Navi Mumbai, India, for the last 17 years, His total Industry experience is 32 +yrs with major Multinationals companies.
Prior to joining Glenmark, he has worked with major multinationals like Hoechst Marion Roussel, now Sanofi, Searle India Ltd, now RPG lifesciences, etc. He has worked with notable Scientists like Dr K Nagarajan, Dr Ralph Stapel, Prof S Seshadri , etc, He did Custom Synthesis for various multinationals in his career like BASF, Novartis, Sanofi, Pfizer etc., He has worked in Drug Discovery, Natural products, Bulk Drugs, Generics, Intermediates, Fine chemicals, Neutraceuticals, GMP, Scaleups, Pharma Plant, API plant etc, he is now helping millions, His friends call him worlddrugtracker.
His New Drug Approvals, All about drugs, Eurekamoments, Organic spectroscopy international, etc in Organic/ Medchem are some most read blogs. He has hands on experience in initiation and developing Novel routes for Drug molecules and implementing them on commercial scale over a 30 year tenure till date Feb’ 2018, Around 30 plus commercial products in his career. He has good knowledge of IPM, GMP, QbD, Regulatory aspects, Technology transfer, Manufacturing, Formulations, Spectroscopy, Stereochemistry, Synthesis, Polymorphism etc. He has several International patents published worldwide.
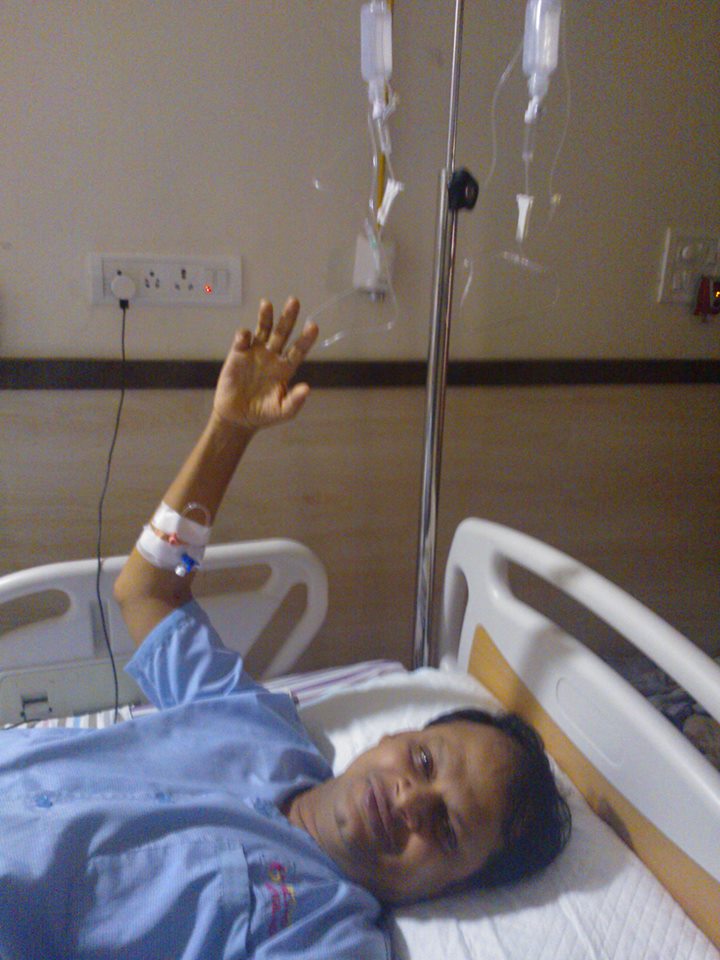
He suffered a one in a million disease in the form of a paralytic stroke/ Acute Transverse Mylitis in Dec’ 2007 and is 90 % paralysed, He is bound to a wheelchair, this seems to have injected feul in him to help chemists all around the world, he is more active than before and is pushing boundaries, he has several million hits on Google, 100 Lakh plus views on dozen plus blogs, 10 Lakh viewers in usa alone, He makes himself available to all, contact him on +91 9323115463, email [email protected], Twitter, @amcrasto
He is a prolific presenter and is invited to major conferences in Mumbai, where he can travel easily. He speaks at universities on topics of Drug discovery, Patents, Qbd, GMP, Tech transfer, polymorphism, Literature search tools, Computer programs and Topics of interest to Pharma Students.
His extraordinary skill on the Computers give him the edge to write/present his thoughts. He demonstrates them to students and professionals alike.
Notably he has 38 lakh plus views on New Drug Approvals Blog in 226 countries, This blog has 6 lakh viewers in USA alone.
Awards
"100 Most Impactful Health care Leaders Global listing", conferred at Taj lands end, Mumbai, India on 14 Feb 2017 by World Health Wellness congress and awards
“Best Worlddrugtracker “ Award for lifetime achievement in Pharma... 7 th July 2017 , The venue...Taj land ends, Bandra, Mumbai India
“Lifetime achievement award” WORLD HEALTH CONGRESS 2017 in Hyderabad, 22 aug 2017 at JNTUH KUKATPALLY. HYDERABAD, TELANGANA, INDIA,
" Lifetime Achievement Award", at the The Middle East Healthcare Leadership Awards - 12th October, 2017 -The Address, Dubai Mall, Dubai...UAE..........Mohammed Bin Rashid Boulevard, Downtown Dubai - Dubai - United Arab Emirates
International award for Outstanding contribution in Pharma at World Health and wellness Congress award, 14th Feb, 2018, at Taj Lands ends, Bandra, Mumbai, India
Conferred very prestigious IDMA award for contribution to society in Pharma at INDIAN DRUGS ANNUAL DAY 2018 VMCC IITBombay Powai, Mumbai India 22 Feb 2018,I was Guest of honor at and was felicitated by president,Indian Drug manufacturers association (IDMA)
Award conferred The Golden Globe Tigers Awards 2018, for Excellence in Pharma, at kuala lumpur, Malaysia, 23 april 2018 at Pullman hotel.
Conferred prestigious individual national award at function for contribution to Pharma society from Times Network, National Awards for Marketing Excellence ( For Excellence in HEALTHCARE) | 5th July, 2018 | Taj Lands End, Mumbai INDIA
Conferred award for Excellence in Pharma by CMO ASIA Award 31st July - 1st August, 2018 | Le Méridien Singapore, Sentosa
Life time Achievement Award by Bioleagues at 4th World Summit on Pharmaceutics Drug Designs, Dubai, UAE. Sep 21-22, 2018 at Flora creek Dubai UAE
Conferred prestigious ABPnews award for "Outstanding contribution to Education Sector" at event ABP News Presents Healthcare Leadership Awards 26th November, 2018 at Taj Lands End, Mumbai India
Honoured to get Award by ETnow for Outstanding contribution to social service at ET Now Presents World Mental Health Congress Co - Hosted with World Health & Wellness Awards, 15th February, 2019. | Taj Lands End, Mumbai, India
Honoured to get prestigious "SPER Innovative Researcher Award" The award was given in the SPER 8th Annual International Conference & Exhibition [SPER 2019] at VYWS’S Inst of pharma education and Research, Wardha [Maharashtra] India on February 23, 2019
Life time Achievement award at BioLEAGUES Worldwide at the 5th International Conference on Pharmaceuticals, API & Mfg. with the theme Enabling Technologies for API, Formulation & Manufacturing Aston Priority Simatupang Hotel & Conference Center, Jakarta, Indonesia on March 15th, 2019.
Lifetime achievement award, Prisal, Bhopal MP, India 26 may 2019 National Pharma Conference organized by Prisal, Pharmaceutical Royal International Society on Pharmaceuticals: Bench to Bedside: Challenges, Recent Initiatives & Future Perspectives
venue, State level animal husbandry auditorium, Bhopal
Honoured to be conferred LIFE TIME ACHIEVEMENT AWARD by LOQMAN AWARDS ( National & International Awards ) on 23rd June 2019 Hotel Saffron leaf, Dehradun, Uttarakhand , India
Life time achievement award, conferred at Bellemie presents Golden Global Health and Education International awards, India Islamic cultural center, Lodhi road, New Delhi, India 7th Sept 2019
Honoured to be conferred Outstanding contribution to Healthcare and Social cause, at “Global Healthcare & Medical Tourism Conclave” 4 th Oct 2019, at ITC Maratha, Mumbai, India,
Lifetime achievement award, conferred at Hotel Flora creek, Dubai, UAE
Organized by Bellemie presents Golden Global Health & Education INTERNATIONAL Conference & Awards 🥇 2019 held on 16th November 2019 at Hotel Flora Creek Dubai UAE
LIFETIME ACHIEVEMENT AWARD conferred by ABP News at ABPNEWS Presents Health Care Leadership Awards 20th November 2019 at Taj Lands End, Mumbai India, Award given by Mr Shailesh Hedge of ABPNEWS, Mr Daara patel, IDMA, Mr Sudarshan jain, IPA, Dr. R L Bhatia Chief Editor & Founder @World CSR Day; Dr Tarita Shankar Chief Managing Trustee SCES
Conferred most prestigious International award at INTERNATIONAL ICONIC AWARD'S 2019 SEASON - 5, Entitled "International lconic Excellent Contribution To Social Services and Education" at The Club, DN nagar , Andheri west, Mumbai India 25th Nov 2019
Thanks to M Nagaman and Nisha rawal for giving the honor
Life time achievement award and Guest of honor status at *Operant Pharmacy Federation* Presenting *World Congress on Pharmaceutical Sciences* on 02-03 December 2019, at Bengkok, Thailand 🇹🇭 with the theme *Empowering New Era Technologies in Pharmaceutical Sciences* Venue: Avani Atrium Bangkok
1880 New Petchburi Road, Bangkapi, Huay Kwang, Bangkok, Thailand
Conferred ETNOW news presents 101 most fabulous healthcare leaders, Global listing at Tajlands end, Bandstand, Bandra (W), Mumbai 400 050, Mumbai, India, 14th Feb 2020, Honour by world health and wellness congress
WOCKHARDT FOUNDATION , SIR DR HUZ KHORAKIWALA TO ANTHONY CRASTO , CONFERRED TITLE AS “WORLD PEACE AMBASSADOR” 20th March , Friday
Dr. Anthony Melvin Crasto Glenmark is now world peace ambassador
SAS Lifetime achievement award 2020 by SAS society, SAS Society has announced "SAS International Awards 2020". #SAS Life time Achivement Awardee 2020. http://www.sassociety.com/sas-conference-and-awards-2020/ Exploring Scholars to Success, ANNOUNCEMENT: SAS INTERNATIONAL AWARDS 2020
Guwahati, Assam, August 03, 2020
WOCKHARDT FOUNDATION AWARD AT ORCHID MUMBAI INDIA 28 JAN 2021
World peace Ambassador Conferred by DR Huzaifa Khorakiwala executive director Wockhardt
I was physically present to take the award,
CONFERRING ON YOU TITLE AS “WORLD PEACE AMBASSADOR”, The Orchid Hotel Mumbai Vile Parle
70-C, Nehru Rd, near Mumbai Domestic airport, Navpada, Vile Parle East, Vile Parle, Mumbai, Maharashtra 400099.
https://goo.gl/maps/vgTNYHwQr3AU2VnF9
Asia pacific excellence award. Title is Life time achievement award
Belgave education society. Commonwealth international university
Gera global education, Research alliance. 30th may 2021 Hotel Radisson Blu. New delhi. Rescheduled to 11th July 2021
Life time achievement award, cooor 27 aug 2021, Holiday inn, mumbai, India
Physical presence event. Award function 27 aug 2021 Award for "Life time achievement award" by Cooor. Council for Brand business promotions and Research in collaboration with News1 India and MSME chamber of commerce and industry 27th aug 2021 at Holiday Inn. Mumbai India
Award received online and speedpost, BHARAT SHIKSHA GAURAV PURASKAR
said prestigious “BHARAT SHIKSHA GAURAV PURASKAR” and a “CERTIFICATE OF EDUCATION EXCELLENCE”. KTK Outstanding Achievers and Education Foundation in association with Helium production
Sunday Nov 14 2021 as virtual event in New delhi
Online 20th Feb 2022 New Delhi India
Lifetime achievement award recieved today from ktk foundation, Life time achievement award 2022
KTK Outstanding Achievers and Education Foundation in association with Helium Production presents lifetime achievement award for industry academic interaction in education, Virtual ceremony 20th Feb 2022 New Delhi
Life time achievement award, At Indore 2nd April 2022 Pharmanest, Operant Pharmacy Federation,
International Conference On Post Covid Era, An International conference on pharma at Radisson Blu Indore india
Life time achievement award from Hon Dr Kiran bedi IPS Governor of puducherry
I was Special guest too at Brand council ratings Conclave and awards 2022 at Holiday Inn, Mumbai India on 16th April 2022, In association with News india 0ne, The Most Prestigious Healthcare Summit " 4th Edition- South Asia Pacific Healthcare Summit and Business Awards 2022", Www.medilinksindia.com/awards
'Top 10 Prominent & Great Personalities of the year 2022' campaign by Fame Finders. 16 JAN 2023
Feeling great to be selected in the 'Top 10 Prominent & Great Personalities of the year 2022' campaign by Fame Finders. It is an online campaign to honour inspiring personalities and feature them in the upcoming edition of the top news sites, including - Deccan Chronicle/Asian Age/Deccan Herald, ANI, Zee5, Latestly, Lokmat Times, DailyHunt, Google News, JioNews, MSN and 70+ sites. LINK www.deccanherald.com/brandspot/pr-spot/top-10-prominent-and-great-personalities-of-the-year-2022-announced-by-fame-finders-media-1181964.html
Proud to recieve Indira Diversity award 2023 for never give up attitude in life, 21 Jan 2023. Indira institute Pune India, Indira Diversity Awards-2023", to witness the presence of super achievers who have contributed to our country's glory, along with organizations that have celebrated inclusivity and diversity,
Weece Global Awards 2023, Feb 15 2023, Honoured to get Lifetime achievement award, Hyat Centric New Delhi India, At GlobalPHT 2023
Lifetime achievement award Conferred by Advantage India Chemical Conclave 2024
Feeling good and thankful
ADVANTAGE INDIA CHEMICAL CONCLAVE 2024, 23 FEBRUARY 2024
HOTEL KOHINOOR CONTINENTAL. Mumbai India
Awarded CASMB FELLOW AWARD at
FMCG Business Conclave Event
1st March 2024
By Pallavi Darade IAS Mh India
Conference Hall No. 105 (A & B), 1st Floor Jio Convention Center, BKC, Mumbai India
Biography
READ MY BIOGRAPHY..........http://scijourno.com/2017/01/09/dr-anthony-crasto/ Written by Mr. Amrit B. Karmarkar
Director, InClinition
SPECIAL CAUSE
15-20 lives saved in corona second wave
Proud to say the my groups 25 nos in total on whatsapp particularly elite (15gps, 3500 members) owners ceo, vp, directors etc
and 10 groups (2500 members) of other group of middle level mgmt, helped people in corona 2nd wave and on record minimum 15-20 lives saved.
Expertise in the area of Process R&D (Generic API)
Development of new API for fresh DMF / CEP Filings for Regulated Markets (US, EU, Canada, Japan etc,).ü Expertise in Lab Development of New API for fresh DMF Filing for regulated markets. Execution of pre-scale up, scale-up and exhibit/validation batches of the new API. Technology Transfer from lab scale to kilo lab, Pilot plant scale and finally to plant scale for commercial Production. Process validation at plant scale to file DMF for regulated markets.
Hand on experience in the Cost reduction / Technology improvement of existing DMF filed APIs by Identifying areas of improvement for existing commercialized processes in all divisions with the objective of “increasing throughput, reducing the steps, recovering and recycling of spent solvents, minimizing waste, effluent generation, reducing cycle time, reducing the overheads corrections, recycles and improve process yield, and RM consumption, using cheaper RM’s, by changing the reaction scheme etc.,"
Expertise in plant level trouble shooting and also in trouble free technology development and Technology Transfer of API.
Expertise in Synthesis of impurities and impurities carry over study/ Genotoxic impurities evaluation / Addressing Regulatory / customer deficiency queries.
Well versed with ICH Guidelines, cGMP, USFDA & Regulatory requirements for various advanced markets like EU, US and Japan, also the regulatory requirements for fresh DMF filing and variation filing of existing process with FDA on existing process. Well versed on the DRA requirements on variation filing of the Technology Improved / cost reduction of existing / commercialized / DMF / CEP filed process.
Well versed with Para – IV Filing, FTF and also regulatory requirements for Drug Substances.
Articulate, dynamic leader with diverse blend of Process R&D / Process Improvement / Process development, tech transfer, Scale–up expertise of API.
|
I stay in Thane, near Mumbai, India
|
|
|
BELOW WITH MY SON LIONEL CRASTO
CLICK ON ICONS BELOW TO CONNECT WITH ME
|
|
Recent News7 lakh views on New Drug Approvals Blog..................click here
1.5 LAKH CONNECTIONS WORLDWIDE 8 MILLION HITS ON GOOGLE |
Subscribe |